Chemical Composition of Wool with Polymeric Structure
Introduction
In this article, I will present chemical composition of wool. I will present polymeric structure of wool polymer. Here, I will present different types of chemical bond of wool fiber. Before going to the main point, at first we will know about wool fiber. As a textile raw material, wool fibers have been used in apparel and home textiles for decades. As well as being used in fashion, wool is increasingly being used in technical textiles. Wool has traditionally contributed significantly to the economies of producers and manufacturers.
The market continues to be dominated by high-value wool products, including carpets, suits, and sweaters. For the last 25 years, the production of clean wool fiber worldwide has been around 1.2 million tons annually, reducing the importance of wool in the textile industry because of man-made fibers dominating the traditional wool market. Conversely, increasing consumer interest in natural, renewable and sustainable materials presents a great opportunity for wool’s future. The quality and yield of wool have also improved significantly over the past few decades. Another significant development in wool science and technology is the use of poor and waste wool for technical purposes.
Chemical composition of wool fiber
The chemical composition of wool fiber leads to a deeper understanding of its remarkable durability, warmth, and versatility. Wool fiber is made from cross linked proteins called keratin. Although keratin makes up the majority of wool fiber, there are also other components included in it. Chemical composition of wool fiber is given below:
| Components | Percentage |
| Keratin | 33% |
| Suint | 28% |
| Fat | 12% |
| Mineral Matter | 1% |
| Different impurities | 26% |
The keratin of wool fiber contains the following basic elements:
| Elements | Percentage |
| Carbon | 50% |
| Hydrogen | 12% |
| Oxygen | 10% |
| Nitrogen | 25% |
| Sulphur | 3% |
Polymeric structure of wool
Wool polymer or polymeric structure of wool is extremely complex. A wool fiber is a cross-linked keratin protein which contains up to 20 different amino acids (glycine, alanine, valine, leucine, isoleucine, phenylalanine, proline, serine, threonine, tyrosine, aspartic, glutamic, arginine, lysine, histidine, tryptophan, cysteine, methionine), making it extremely complex. A polypeptide is an amino acid that bonds with another amino acid. A peptide bond is the bond between two amino acids from protein polymers.
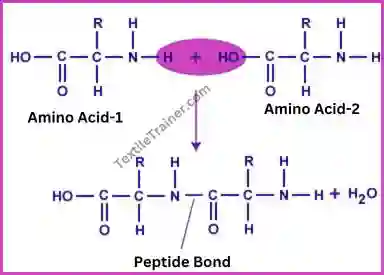
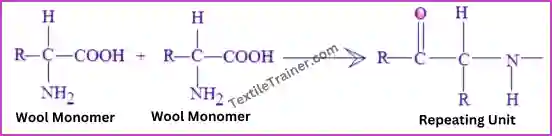
A wool polymer (Polypeptide chain) is formed by linking amino acids together with a peptide bond (-CO-NH-). The repeating unit of wool polymer in the figure is:
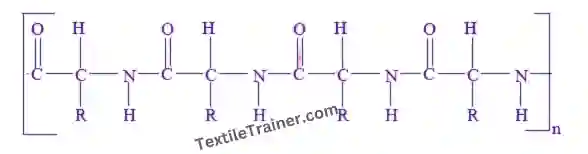
The wool polymer is combined of amino acids in long polypeptide chains, as a linear keratin polymer with some very short side groups and it has a helical configuration. Each variety of wool contains a different amino acid sequence and content. In wool protein chains, the disulfide cross linked cysteine, a di-amino acid contained in two adjacent chains, is periodically used to join the chains. About 40% of protein chains form helixes by spiraling upon themselves and hydrogen bonding internally.
Spiral or Helical structure of wool
In its relaxed form, keratin has a spiral structure called alpha-keratin, responsible for wool’s high elongation. When the wool stretch, the polymers unfold into a beta-keratin structure.
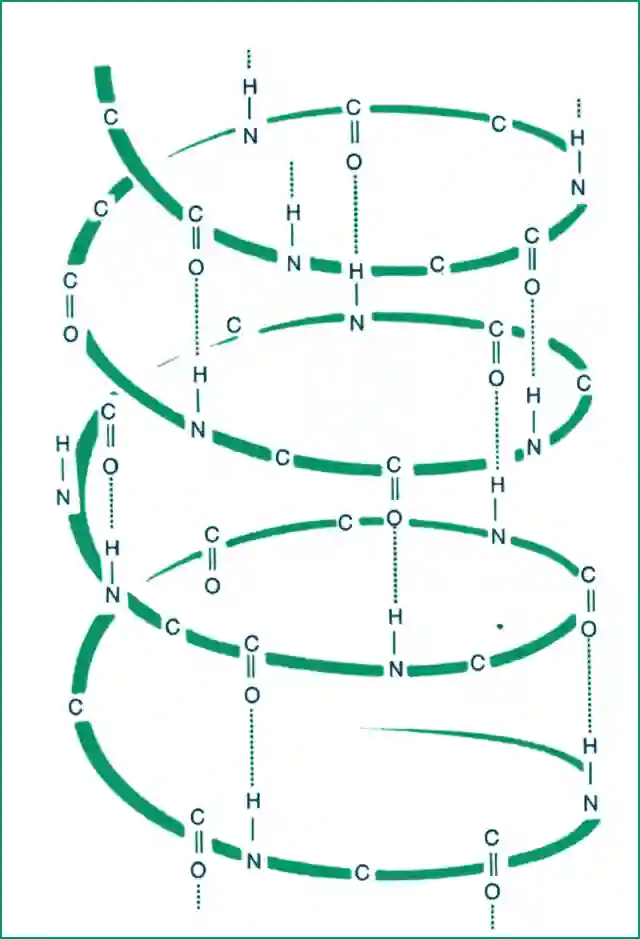
Wool’s tenacity is enhanced by hydrogen bonds between oxygen and hydrogen atoms in alternate spirals of the helix. This strengthens the structure and requires greater force to stretch them.
Types of Chemical bonds of wool
In wool fiber, keratin is a cross-lined protein. It also found in horns, hooves and in human hair and fingernails. Keratin made up of carbon, hydrogen, oxygen, nitrogen, and sulfur that combine to form 17 amino acids. The amino acids shown in table: 01. A natural cross-link keeps the flexible molecular chains of wool together. The cross links in wool polymers are hydrogen bonds, cystine or sulfur bonds, ion-to-ion bonds called salt bridges, peptide, ester, and ether bonds. There is also van der waals force present in wool polymer.
- Cystine linkages: Cystine links are the most important part of the molecule. Any chemical, such as alkali, that damages this linkage can destroy the entire structure. To finish wool, the linkage can be broken and then reformed. Pressing and steaming can make minor modifications to the cystine linkage. Incorrect washing and exposure to light can destroy the links and damage the fiber. The cystine linkages contribute to lateral resistance, strength and cysteine linkages also react with alkali, bleaches, heat, and sunlight.
- Hydrogen bonds: The hydrogen bonds provide strength, elasticity, temporary strength to the cell, and when they react with moisture they debone some intermolecular forces and decrease the strength of the cell.
- Salt bridge: It contributes to strength as well as reacting with acids and dyes to form a strong substance.
Amino Acid residues in wool fiber
| Amino acid | Structure of side chain (R ) | Characteristic of side chain | Amount g/100g of protein |
| Glycine Alanine Phenylalanine Valine Leucine Isoleucine | H- CH3– C6H5-CH2– (CH3)2-CH – (CH3)2-CH-CH2 – CH3-CH2-(CH3)CH- | None polar | 6.5 4.4 3.7 4.7 11.3 1.4 |
| Serine Threonine Tyrosine | HO-CH2– CH3-CH(OH)- HO-C6H4-CH2– | Hydroxyl containing | 9.4 6.8 5.8 |
| Methionine Cysteine | CH3-S-CH2-CH2– -CH2-S-S-CH2– | Sulfur containing | 0.7 12.7 |
| Lysine | NH2-CH2-CH2-CH2-CH2– | Basic | 3.3 |
| Aspartic acid Glutamic acid | HOOC-CH2– HOOC-CH2-CH2– | Acidic | 7.3 15.3 |
You May Read:
- History of wool with geographical distribution.
- Classification of wool fiber.
- Morphology of wool fiber.
- Differences Between Wool and Worsted Fabric.
- Sizing Easy Calculation.
Reference:
- Cook, J. G. (2005). Handbook of Textile Fibers. Delhi: WoodHead Publishing Limited.
- Corbman, B. P. (1987). Textile Fiber to Fabric. Singapore: McGraw Hill Book Co.
- Dr. Hosne Ara Begum, P. D. (n.d.). Natural fibers. Dhaka: Hafiz book center.
- Kaplan, N. (2006). Textile Fibres. New Delhi: Abhishek Publications.



