Easy Application Direct Dye on Cotton & Recipe calculation
Name of the Experiment:
Lab report on dyeing process of cotton fabric with direct dyes/ study on direct dye on cotton with recipe calculation.
Introduction:
In 1883, J. Walter of Geigy discovered the first synthetic direct dye, which was Direct Yellow 11. In 1956, the Society of Dyers and Colorists (SDC) defined direct dyes as “anionic dyes substantive to cellulose when applied from an aqueous bath containing electrolytes”. Direct dyes are applied to cellulosic fibers by adsorption, diffusion, and migration. It is the structure, morphology and use of electrolytes that most influence the dyeing mechanism of cellulose fiber.
When the cellulosic fiber is immersed in water the amorphous regions of the fiber swell to create small pores in the order of 20 – 100 A (Angstrom) units – these pores allow dye molecules of smaller size to diffuse into the fiber structure. The addition of electrolytes (e.g. sodium chloride, sodium sulfate) facilitates diffusion and exhaustion of direct anionic dye by neutralizing the negative surface charge of cellulosic fibers. As dye anions move from dye-in-solution to dye-in-fiber, hydrogen bonding and van der wales forces bind the dye anions to the fiber.
Objectives:
- To know about direct dyes.
- Come to know application parameters of direct dye.
- To know working procedure of dyeing process of direct dye.
A direct dye is a versatile dyestuff that can use on fibers such as cellulose, wool, silk, and nylon. Although the direct dyes have deficiency in wet colorfastness, they exhibit a broad range of fastness properties, as shown by their use in apparel, upholstery, lining, and automotive fabrics. For direct dye exhaust dyeing to be successful, it is important to consider the following application parameters:
- Temperature should be around 1000 C.
- pH should be 7-9 in case of cellulosic diber.
- Electrolyte concentration should be (<0.5% shade=5 gm/l, 1.0-2.0% shade= 10-20 gm/l, 2.0-4.0% shade= 20-30 gm/l, >4.0% shade=30-40 gm/l)
- Dye bath additives.
Typical recipe for Direct Dye on Cotton:
| Chemical | Amount(gm/l) |
| Wetting agent | 1.0-2.0 gm/l |
| Sequestering agent | 1.0-2.0 gm/l |
| Leveling agent | 0.5-1.0 gm/l |
| Soda ash | 1.0-5.0 gm/l |
| Salt | 20-30 gm/l |
| Direct dye | 2% |
| Temperature | 950 C |
| Time | 30-50 min |
| pH | Neural to alkaline |
| M:L | 1:20 |
| Sample weight | 5 gm |
Recipe Calculation:

Process Curve:
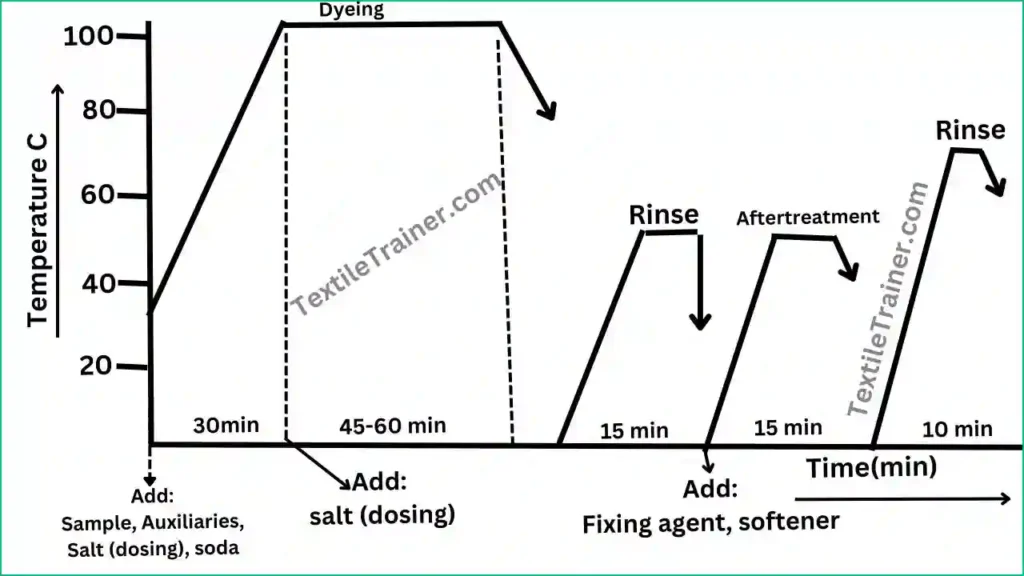
Working procedure:
- At first, set the dye bath with the sample at room temperature.
- Now, add dye solution with others chemical and auxiliaries. Raise the temperature to 900C.
- Run the dye bath for 15-20 min and add salt step by step according to dye bath concentration. If need higher depth shade, need more salt concentration.
- But, it is better to add this salt after reaching the temperature to boiling point. Because at boiling point, fiber become more swell. At this period the maximum dye molecule penetration to the sample.
- Run the dye bath for 30-50 min at 90-950 c for complete the dyeing cycle.
- Now, cool down the temperature to 60-700C.
- Drop the bath liquor and rinse the sample.
- As we know, fastness properties of direct dye is not good. After dyeing, sample need to after treatment to improve the fastness properties.
After treatment process:
A suitable fixing agent is used after treatment to improve color fastness properties; usually, this process is at 30-40 C. Occasionally, 60 C is used for 15-20 minutes or according to the seller’s recommendation. After the last rinse bath, a cationic softener is added to the fabric to enhance its handle properties; this process is performed at 40-50C for 15-20 minutes.
Result:

Conclusion:
As we know, direct dyes are some of the most versatile dyestuff that can be applied to cellulose, wool, silk, and nylon fibers. We applied direct dyes to cotton fabric in this experiment. We learned from this experiment how to calculate the recipe and how to work the direct dye on cotton fabric. This will be helpful for us in the future.
More Lab Report: