Combined Scouring and Bleaching Process of Wool Fabric
Name of the Experiment
Study on Combined scouring and bleaching process of wool fabric.
Introduction
The scouring of wool Raw wool contains a variety of different natural impurities, including wool wax (18%), suint (10%), water (12%), as well as vegetable and mineral debris (15°/0). The wool fibers are sensitive to strong alkalis, so it is often treated with mild alkaline conditions at an appropriate temperature since they are affected by strong alkalis. The wool readily dissolves when it treats with a strong alkaline solution, especially at high temperatures. At boiling temperatures, a 3% solution of sodium hydroxide will dissolve wool completely. Using alkalis to treat wool doesn’t adversely affect its mechanical properties; however, it can cause yellowing, which is determined by the time of the treatment, the temperature and the concentration of alkali solution used.
In contrast, bleaching wool with hydrogen peroxide is a common method for bleaching wool nowadays. It is also possible to do this by using other methods like stoving (a method of using gaseous sulfur dioxide) or soaking (a method of applying acidified sodium bisulfite to a cloth). The problem with both of these processes is that sulfur dioxide is generated during this process and reacts with water in order to form sulfuric acid. As with garment dyeing, bleaching occurs within a very short time.
Objectives
- To know scouring and bleaching process of wool fabric.
- To know standard recipe for scouring and bleaching process of wool fabric.
- Come to learn recipe calculation of scouring and bleaching process of wool fabric.
- To know working procedure of combined scouring and bleaching of wool fabric.
Typical Recipe for Combined Scouring and Bleaching Process of Wool
| Chemical | Amount (gm/l) |
| Wetting agent | 0.5-1.0 gm/l |
| Detergent | 1.0-2.0 gm/l |
| Peroxide stabilizer | 0.5-1.0 gm/l |
| Sodium tri-polyphosphate/ammonia | 2-3 gm/l |
| Hydrogen peroxide (35%) | 10-20 gm/l |
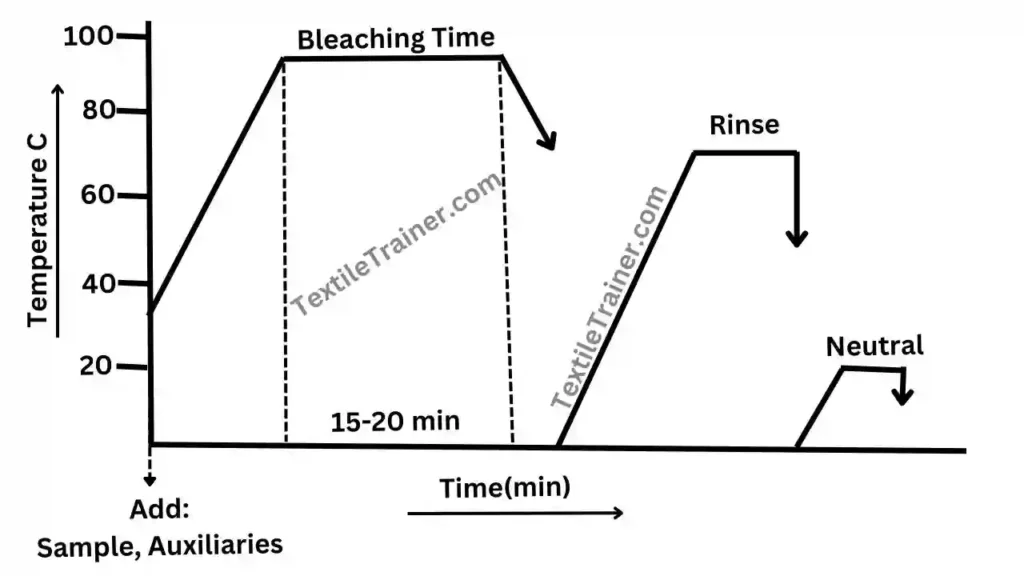
| Temperature | 70-800 C |
| Time | 15-20 min |
| pH | 8.5-9 |
| M:L | 1:20 |
| Sample Weight | 5 gm |
Recipe Calculation

Process Curve
Working procedure
- At first, set the bath with wetting agent, detergent, peroxide stabilizer, sodium tri-polyphosphate or ammonia at room temperature.
- Rise the temperature to 500C and add hydrogen peroxide. Now rise the temperature to 70-800 C with the rate of 1-20C/ min.
- Treat the sample for 15-20 min, after reaching the temperature to 70-800 C.
- Then, cool down temperature and drop the liquor.
- Rinse twice with hot and cold water successively.
- After rinse, sample treat with the per-oxide killing agent to remove residual per-oxide from the sample.
- Finally, we squeeze the sample and dryer the sample.
Result

Precaution
- Wool fiber is very sensitive to alkali. So mild alkali should be used.
- Be careful when calculate the required chemical amount.
- Used safety glass and gloves when measured the chemical.
Conclusion
It is known that wool fiber is sensitive to alkali. That is why the combined process of scouring and bleaching is done in a very short timeframe. Mild alkali should be used in case of wool pre-treatment process. Strong alkali like caustic soda can completely destroy wool. However, this experience will be helpful in our future lives. Thanks to our teacher for helping us with this experiment.
Reference:
- Belal, P. D. (2016). Understanding Textiles for a Merchandiser. Dhaka: LB Graphics & Printing.
- Hossain, M. F. (2015). Practice of Textile Coloration, Volume-I. Dhaka: Books Fair Publications.
- Kabir, D. S. (2016). Chemistry of Dyes & Pigments. Dhaka: Books Fair Publications.
- Kadolph, S. J. (2006). Textiles. New Delhi: Pearson Education.
- Cook, J. G. (2005). Handbook of Textile Fibers. Delhi: WoodHead Publishing Limited.
More Lab Report: